GenomeScan. Your partner in Genetic Research and Diagnostics.
We offer genetic analysis for laboratory research and patients.
We offer genetic analysis for laboratory research and patients.
We support scientific community of pharmaceutical industry, biotechnological and agricultural companies, healthcare providers and academic institutions, that have an impact on Healthcare and Quality of Life. To assist you with your mission, we leverage innovative Next Generation Sequencing (NGS) technologies to advance solutions from basic research through preclinical stage to every-day clinical practice, paving the way towards personalized medicine.
We provide tailored, end-to-end solutions. Our experienced team guides you along every step: from designing your experimental setup, through sample preparation and full stack reporting, to interpretation of your data with a turnaround time that fits your needs.
Our passionate team consists of scientists and technicians with diverse scientific backgrounds. You can consider them as an extension of your own team with a commitment to provide you with excellent, robust, and fast NGS services with matched bioinformatics solutions.

With a track record of nearly two decades in genetic services and strong support and shareholding of large Academic Medical Centers, we are a partner you can rely on. Whether it involves batched or single-sample processing, prospective or retrospective clinical cohorts, archive samples, biosafety testing material, plant, or microbiome samples, we will deliver.

Our well-established contacts with technology providers, our collaborations with academic researchers, and out participation in numerous scientific consortia spark our innovation to grant you access to the most advanced sequencing methodologies and solutions.
We deem quality of a paramount importance, and we fully embrace it by operating under ISO/IEC 17025 and ISO 15189 accreditation. Your personal data and privacy are secured via data management system that guarantees the protection in compliance with (inter)national directives.
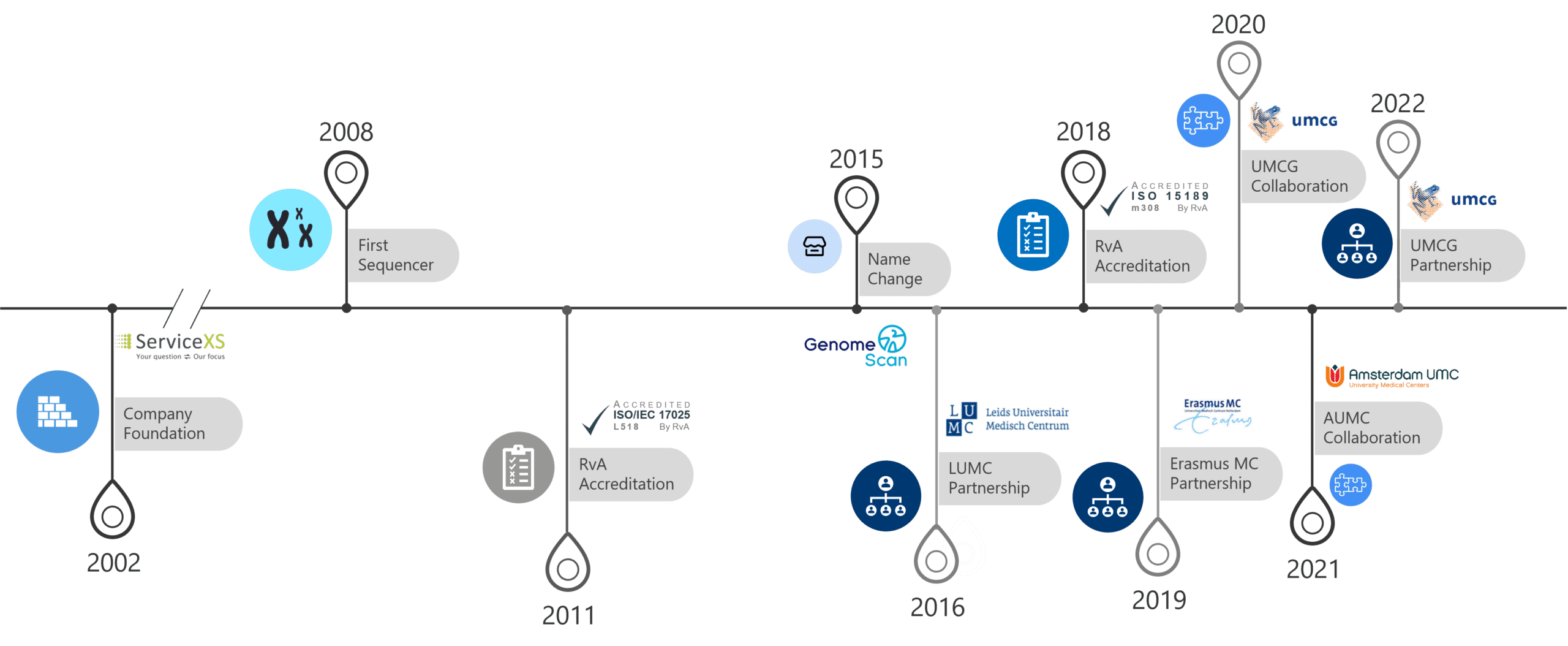
GenomeScan is grounded in a combination of scientific partnership, technology innovation, and leadership experience. Our founders believe that Next Generation Sequencing applications can contribute to improve diagnosis of genetic disorders, ensure better disease risk management, and accelerate access to personalized medicine.
GenomeScan is engaged in developing new tools to diagnose genetic disorders more quickly, more affordably and more effectively. The company fosters product innovation through partnership with medical centers and research laboratories.
Three times a year, GenomeScan comes together with the Scientific Advisory Board, an indispensable factor for our success and a great source of inspiration. The board consists of accomplished scientists coming from diverse fields of research and academic institutions who provide us with intellectual, scientific and strategic support. Membership is comprising of:
“attending pathologist” and “registered clinical molecular scientist in pathology” at the Department of Pathology, LUMC, Leiden
Professor Morreau furthermore works on the familial basis of colorectal cancer, on endocrine tumors and the refinement of diagnostics using molecular insights.
Head of Human Genetics department, LUMC, Leiden.
The scientific interests of professor van der Maarel concentrate on the genetic and epigenetic regulation of repetitive DNA in the human genome in relation to disease.
Clinical Geneticist, Department of Clinical Genetics, LUMC, Leiden
Dr. Santen performs, apart from his clinical duties, clinical and molecular research into Coffin-Siris syndrome and other SWI/SNF related diseases. He also investigates broader application of Next Generation Sequencing technology to identify potential causative mutations in urgent situations, for example during pregnancy when ultrasound anomalies are detected, and shortly after birth in the case of children admitted to the ICU.




Please either fill in this form or email us directly at info@genomescan.nl
and we will get in touch with you to discuss your requirements.